If you’ve come across the term HCOOCH CH2 H2O in a chemistry textbook, lab report, or research paper, you’re probably wondering what it actually means and why it matters. This combination of chemical notation touches on ester chemistry, hydrolysis reactions, and some genuinely useful real-world applications. Whether you’re a student brushing up on organic chemistry or a curious reader who loves science, this guide breaks it all down in plain language.
What Does HCOOCH CH2 H2O Actually Mean?
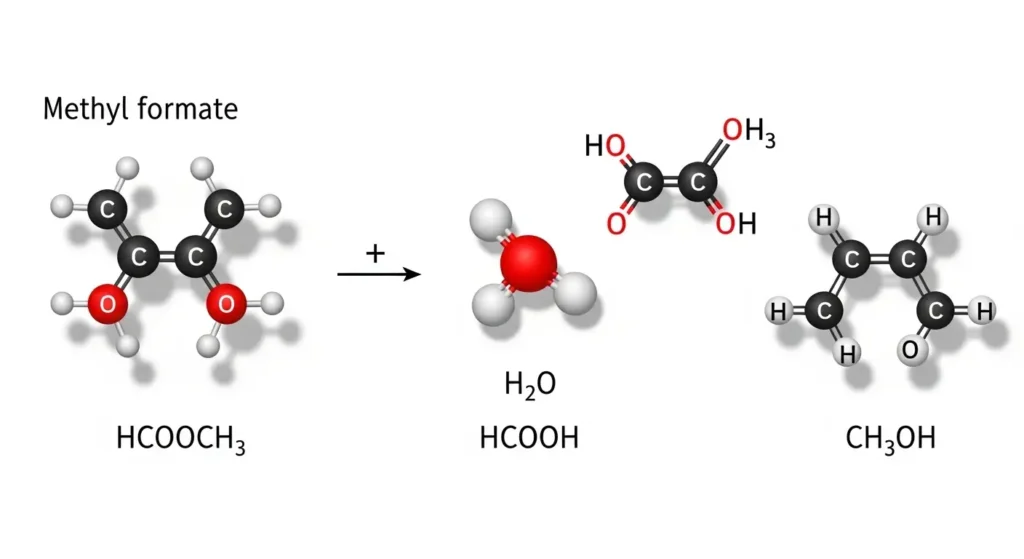
Let’s start from the beginning. HCOOCH is the chemical representation of a formate ester group — specifically, it points to methyl formate or a related formate compound depending on the full molecular context. The CH2 portion refers to a methylene group, a carbon atom bonded to two hydrogen atoms. And H2O, of course, is plain water.
When you put these together in a chemical equation, you’re usually looking at a hydrolysis reaction — a process where water breaks a chemical bond in the ester.
In simple terms:
- HCOOCH = formate ester linkage
- CH2 = methylene bridge
- H2O = water acting as a reactant
Together, they describe how an ester compound reacts with water, typically producing a carboxylic acid (formic acid, HCOOH) and an alcohol.
The Chemistry Behind Ester Hydrolysis
How the Reaction Works
Ester hydrolysis is one of the most fundamental reactions in organic chemistry. When a formate ester like HCOOCH₂ reacts with water (H₂O), the water molecule attacks the carbonyl carbon of the ester. This breaks the C–O bond and produces:
- Formic acid (HCOOH) — the carboxylic acid component
- Methanol or related alcohol — depending on the ester’s structure
The reaction can happen under two conditions:
- Acid-catalyzed hydrolysis — uses an acid like HCl or H₂SO₄ to speed things up
- Base-catalyzed hydrolysis (saponification) — uses a base like NaOH, which drives the reaction to completion
Why Water Plays a Critical Role
Water isn’t just a bystander in this reaction. It’s an active participant. The oxygen atom in H₂O donates electrons and attacks the electrophilic carbon in the ester group. Without water, this reaction simply doesn’t happen under normal conditions. That’s why controlling moisture levels is so important in industries that work with ester compounds.
Battlefield 6 Cheats: What You Need to Know in 2025
Real-World Applications of Formate Ester Chemistry
You might be thinking — okay, but where does this actually show up in real life? More places than you’d expect.
Industrial Uses
- Leather tanning industry — Formate esters and their hydrolysis products are used in processing animal hides.
- Textile manufacturing — Used as a reducing agent and pH regulator in dyeing processes.
- Pharmaceutical synthesis — Formate groups appear as intermediates in drug manufacturing.
- Food industry — Formic acid (the hydrolysis product) is approved as a preservative in some countries.
Laboratory Applications
In a lab setting, reactions involving HCOOCH CH2 H2O help chemists:
- Understand ester reactivity
- Practice nucleophilic acyl substitution mechanisms
- Study the kinetics of hydrolysis under different pH conditions
HCOOCH CH2 H2O in Organic Reaction Mechanisms
The Nucleophilic Acyl Substitution Pathway
This is the core mechanism at play. Here’s a simplified breakdown:
- Water acts as a nucleophile and attacks the carbonyl carbon of the formate ester.
- A tetrahedral intermediate forms temporarily.
- The intermediate collapses, pushing out the leaving group (the alkoxide).
- A proton transfer completes the reaction, giving you formic acid and an alcohol.
This mechanism is the same whether you’re working with simple esters or complex biological molecules like fats and oils.
Acid vs. Base Conditions — What’s the Difference?
Under acidic conditions, the carbonyl oxygen gets protonated first, making the carbon more electrophilic and easier to attack. The reaction is reversible.
Under basic conditions, hydroxide ion (OH⁻) does the attacking instead of water. This drives the reaction irreversibly forward, which is why soap-making (saponification) works so well — you get complete conversion every time.
Pros and Cons of Formate Ester Hydrolysis
Pros
- Simple reaction setup — Doesn’t require exotic reagents or extreme conditions
- Predictable products — You know what you’re going to get (formic acid + alcohol)
- Versatile — Works under both acidic and basic conditions
- Useful byproducts — Formic acid has multiple commercial applications
- Well-studied — Decades of research make this a reliable reaction to work with
Cons
- Formic acid is corrosive — Handling the product requires proper safety precautions
- Reversibility under acidic conditions — You may not achieve full conversion without excess water
- Sensitivity to temperature — Reaction rates vary significantly with temperature changes
- Not suitable for all esters — Bulky or sterically hindered esters react much more slowly
Common Mistakes People Make
Even experienced chemists and students slip up with this chemistry. Here are the most frequent errors:
1. Ignoring stoichiometry Using too little water is a common problem. Since water is a reactant, you need it in sufficient quantity — usually in excess — to push the reaction forward.
2. Confusing hydrolysis with esterification These are reverse reactions. Hydrolysis breaks esters apart; esterification builds them. Students sometimes mix up the direction of the reaction when reading equations.
3. Forgetting the role of pH pH doesn’t just speed things up — it changes the mechanism. Treating acid-catalyzed and base-catalyzed hydrolysis as identical leads to wrong predictions about products and yields.
4. Skipping safety protocols Formic acid, even in dilute form, can irritate skin and eyes. Always work in a fume hood and wear appropriate protective gear.
5. Misreading molecular notation HCOOCH and CHOOCH look similar but represent different structural arrangements. Always draw out the full structural formula to avoid confusion.Best Practices for Working with HCOOCH CH2 H2O Reactions
If you’re running these reactions in a lab or studying them for an exam, keep these tips in mind:
- Always use excess water to drive the equilibrium toward hydrolysis products
- Control your pH carefully — use a buffer if you need consistent conditions throughout the reaction
- Monitor temperature — most formate ester hydrolysis reactions proceed well between 50–80°C
- Use thin-layer chromatography (TLC) to track reaction progress without disrupting the reaction mixture
- Neutralize the formic acid product immediately if you’re sensitive to acidic conditions in downstream processes
- Label everything — formate esters and formic acid look similar in solution but behave very differently
Conclusion
The notation HCOOCH CH2 H2O represents more than just a string of letters and numbers — it describes a meaningful chemical interaction between a formate ester and water. This hydrolysis reaction sits at the heart of organic chemistry and finds applications in industries ranging from pharmaceuticals to food preservation.
Understanding how the ester bond breaks, what role water plays, and how pH shifts the mechanism gives you a solid foundation for tackling more complex chemistry down the road. Whether you’re prepping for an exam or working through a synthesis problem, getting comfortable with formate ester hydrolysis is genuinely worth the effort.
Stay curious, keep drawing out your mechanisms, and don’t skip the safety steps.
Frequently Asked Questions (FAQs)
1. What is HCOOCH in chemistry?
HCOOCH represents a formate ester group. It consists of a formyl group (HCO–) bonded to an oxygen atom, which is then connected to a carbon chain. It’s the ester derived from formic acid.
2. What happens when HCOOCH CH2 reacts with H2O?
When a formate ester reacts with water, hydrolysis occurs. The ester bond breaks, producing formic acid (HCOOH) and the corresponding alcohol. The reaction can be catalyzed by either acid or base.
3. Is formic acid dangerous?
Yes, formic acid is corrosive and can cause skin and eye irritation. In concentrated forms, it’s toxic. Always handle it with gloves, eye protection, and in a well-ventilated space.
4. What is the difference between acid and base hydrolysis of esters?
Acid hydrolysis is reversible and requires a proton donor to activate the ester. Base hydrolysis (saponification) is irreversible because the carboxylate salt formed is stable and doesn’t re-esterify. Base hydrolysis typically gives higher yields.
5. Where is formate ester hydrolysis used industrially?
It’s used in leather tanning, textile dyeing, food preservation, and pharmaceutical intermediate production. Formic acid — the product of hydrolysis — is also used as an antibacterial agent in livestock feed in some regions.